Virus Infects Cells with a Protective Cloaking Mechanism
Discovery of jumbo phage’s stealth compartment could be leveraged to engineer new therapies to treat antibacterial-resistant infections
Published Date
Article Content
In a growing global trend, bacteria have evolved new ways to maneuver around medical treatments for a variety of infections. The rising antibiotic resistance crisis poses a significant public health threat in hospitals and other settings, with infections resulting in millions of deaths in recent years.
Scientists are now looking to bacteriophage — viruses that infect bacteria — and their potential to treat drug-resistant infections. They have begun to look deeper into an intriguing class of large bacteriophage known as “jumbo phage” that exhibit extraordinary features as possible new agents for bacterial infection treatments.
A study led by University of California San Diego researchers has shed new light on the unusual ways that phages have evolved to infect bacteria. Over millions of years, viruses and bacteria have engaged in a back-and-forth arms race. Viruses develop new ways to infect bacteria, while bacteria counter by evolving a resistance mechanism.
In order to fully realize the potential of jumbo phages and their promise as new therapeutics, researchers must decipher the mechanisms they employ to infect bacteria and evade the host’s defenses.
A new study published in the journal Cell Host and Microbe describes the first-of-its-kind discovery of a type of membrane-bound sac, or vesicle, used by jumbo phages of the Chimalliviridae family. A team that includes researchers from the Innovative Genomics Institute at UC Berkeley found that immediately after jumbo phages infect a bacterial cell, they form a structure that shields and hides valuable DNA material. Phages use this genetic material to develop a nucleus inside their bacterial hosts. The newly discovered compartment, which they named the EPI, or early phage infection vesicle, serves as a type of cloaking device that prevents triggering the bacteria’s immune system.
“When phage infect a bacterial cell, the EPI vesicle protects the genome of the virus during early stages of infection when it’s very vulnerable,” said Emily Armbruster, a UC San Diego School of Biological Sciences graduate student and co-lead author of the study. “Bacteria and viruses are often dismissed as simple organisms but they’re actually capable of very sophisticated intracellular warfare and this study is a new example of that.”
The new findings are part of UC San Diego’s advancements in developing innovative therapies using phages, highlighted by a consortium funded by a Howard Hughes Medical Institute's (HHMI) Emergent Pathogens Initiative. Also, the university’s Center for Innovative Phage Applications and Therapeutics (IPATH) is the first dedicated phage therapy center in North America focusing on medical solutions to combat antibiotic resistant infections.
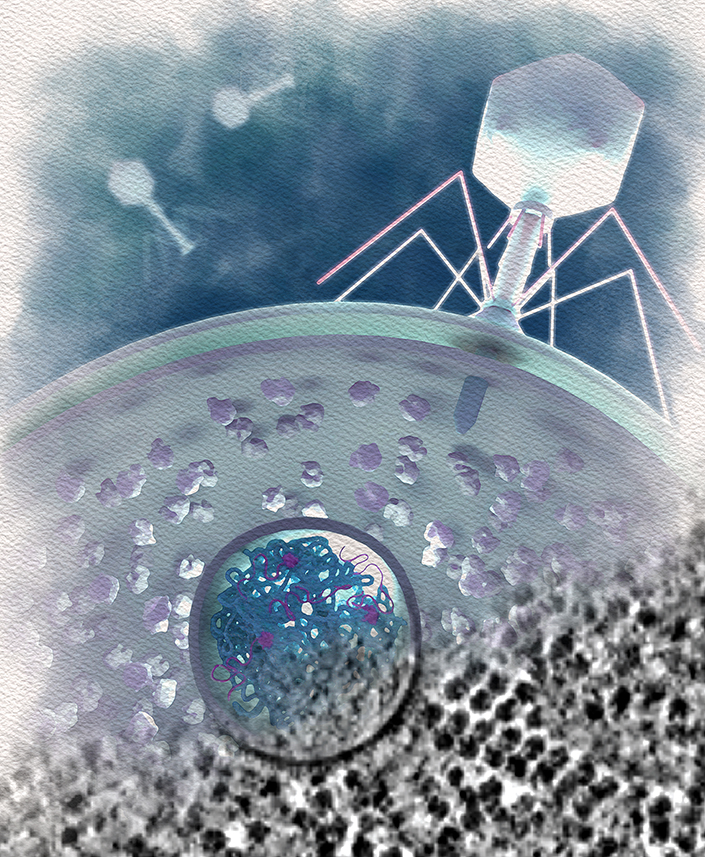
An artistic depiction of a jumbo phage infecting a bacterium, with the EPI vesicle (circle) shown protecting the phage DNA material, which is preparing to form a nucleus by synthesizing RNA (magenta) and protein (gray tails emerging from the EPI). Credit: Margot Riggi

Co-lead authors of the new study Postdoctoral Scholar Phoolwanti Rani (left) and Graduate Student Emily Armbruster.
Because most phages simply inject their DNA directly into the host, effectively announcing their arrival within the cell, the results of Chimalliviridae phage’s stealth approach came as a revelation to researchers.
“The discovery of this vesicle was completely surprising. The bacteria don’t realize that there’s a virus in there, producing things that will eventually take over,” said Joe Pogliano, a professor in the Department of Molecular Biology and a senior author of the paper. “If we have other phages that are really good at infecting bacteria, the hope is that we might be able to give them this vesicle for protection. The goal is to make phages that are better at killing bacteria so that we can have a range of phage therapy applications.”
Armbruster identified curious DNA “dots” within infected cells under a light microscope. Professor Elizabeth Villa’s laboratory then used high-end imaging technologies to discover that these dots were tiny vesicles containing viral DNA and molecular machineries outside these vesicles. Paper co-lead author Phoolwanti Rani, a postdoctoral scholar in the Villa Lab, found that these vesicles were actually metabolically active, confirming the purpose of the molecular machines hanging outside the vesicles.
“These vesicles are one-of-a-kind in bacteria,” said Rani. “Not only did we show that these vesicles are making RNA, but also they are getting ready to establish infection by synthesizing genes important for nucleus formation.”
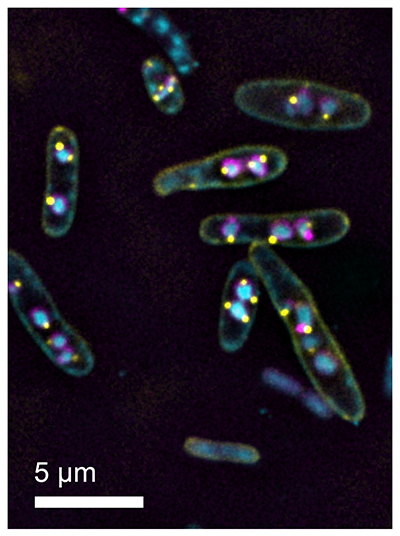
Fluorescence microscopy image of E. coli cells infected with bacteriophage Goslar, with both the phage nuclei and EPI vesicles fluorescently labeled. The viral DNA is inside the phage nuclei (cyan), surrounded by a shell made of phage protein ChmA (magenta). The EPI vesicles (yellow dots) remain attached to the outside of the phage nuclei, having transferred their DNA to the phage nuclei as part of the phage life cycle.
“A few years ago we discovered the phage nucleus that protects the replicating genomes. The question remained: how is the first genome protected?” said Villa, a professor in the Department of Molecular Biology and HHMI Investigator. “Discovering a second compartment that is metabolically active blew our minds, and will be key for therapeutic applications of phage.”
Crucial to the findings was the use of “CRISPR interference by antisense RNA targeting” (CRISPRi-ART), a new CRISPR-based technology recently published in Nature Microbiology from the labs of Brady Cress and Nobel laureate Jennifer Doudna at the Innovative Genomics Institute (UC Berkeley). This work, co-led by postdoctoral scholar Ben Adler and PhD student Muntathar Al-Shimary from the Doudna lab, describes how CRISPRi-ART targets messenger RNA and halts the production of proteins that researchers target for analysis. In the new study from UC San Diego, the researchers used CRISPRi-ART to demonstrate that the EPI and nucleus-forming stages are both necessary for the Chimalliviridae’s normal life cycle and infection process.
The full authors list includes: Emily G. Armbruster, Phoolwanti Rani, Jina Lee, Niklas Klusch, Joshua Hutchings, Lizbeth Y. Hoffman, Hannah Buschkaemper, Eray Enustun, Benjamin A. Adler, Koe Inlow, Arica R. VanderWal, Madelynn Y. Hoffman, Daksh, Ann Aindow, Amar Deep, Zaida K. Rodriguez, Chase J. Morgan, Majid Ghassemian, Thomas G. Laughlin, Emeric Charles, Brady F. Cress, David F. Savage, Jennifer A. Doudna, Kit Pogliano, Kevin D. Corbett, Elizabeth Villa and Joe Pogliano.
Support for the research was provided by a Howard Hughes Medical Institute Emerging Pathogens Initiative grant; the National Institutes of Health (grants R01-GM129245, R35 GM144121 and R01-GM57045); a National Science Foundation Major Research Instrumentation grant (NSF DBI 1920374); InCoGenTEC; m-CAFEs and the Curci Foundation.
Competing interest disclosures: Kit and Joe Pogliano have an equity interest in Linnaeus Bioscience Incorporated and receive income. The Regents of the University of California have patents issued and pending for CRISPR technologies on which Doudna, Cress, Adler, Savage, Charles, Kit and Joe Pogliano, Armbruster and Lee are inventors. Doudna is a cofounder of Azalea Therapeutics, Caribou Biosciences, Editas Medicine, Scribe Therapeutics, Intellia Therapeutics, and Mammoth Biosciences, and is a scientific advisory board member of Vertex, Caribou Biosciences, Intellia Therapeutics, Scribe Therapeutics, Mammoth Biosciences, Algen Biotechnologies, Felix Biosciences, The Column Group and Inari. Doudna also is Chief Science Advisor to Sixth Street, a Director at Johnson & Johnson, Altos and Tempus and has research projects sponsored by Apple Tree Partners and Roche. Savage is a cofounder and scientific advisory board member of Scribe Therapeutics.
Share This:
You May Also Like
Stay in the Know
Keep up with all the latest from UC San Diego. Subscribe to the newsletter today.