Genome-Wide Analysis Reveals New Strategies to Target Pancreatic Cancer
Researchers discover unexpected role of an immune system receptor; blocking it halts human cancer cell growth and improves survival in animal models
Published Date
By:
- Scott LaFee
Share This:
Article Content
For some cancers, initial treatment with chemotherapy brings positive, but only temporary, results: tumors shrink, but then rebound as the cancer becomes drug-resistant. This pattern of remission-resistance-relapse is particularly true for pancreatic cancer, an aggressive disease in which early success is often countered by eventual disease progression.
To wit: The one-year relative survival rate for pancreatic cancer is 20 percent, according to the American Cancer Society. The five-year rate is just 7 percent.
The reason: Current multidrug chemotherapy regimens targeting pancreatic cancer typically do not fully eradicate all cancer cells, leaving behind drug-resistant cells that harbor aberrant stem cell properties and can drive tumor regrowth and metastasis.
In a new paper publishing in the April 4, 2019 online issue of Cell, an international team of scientists led by researchers at University of California San Diego School of Medicine employed an array of next-generation sequencing and gene-editing tools, such as CRISPR, to map the molecular dependencies —and thus vulnerabilities — of pancreatic cancer stem cells.
Most notably, they found that a key hormone receptor called retinoic acid receptor-related orphan receptor gamma or RORγ, previously studied in inflammation and T-cell differentiation, was especially active during pancreatic cancer progression, and that blocking it markedly slowed patient derived tumor growth and improved survival in animal models.
“These studies revealed an unexpected role for immuno-regulatory genes in the maintenance of the most aggressive, drug-resistant cells in pancreatic cancer,” said senior study author Tannishtha Reya, PhD, UC San Diego professor in the departments of Pharmacology and Medicine. In particular, RORγ emerged as a key regulator. RORγ rose with cancer progression and its inhibition through genetic and pharmacological approaches resulted in a striking defect in pancreatic cancer growth.
“Our work shows that immune system signals are hijacked by pancreatic cancer, and suggests that therapies currently being tested for autoimmune indications should be considered for testing in pancreatic cancer,” said Reya.
Using genome scale approaches to map stem cell dependencies in pancreatic cancer “will be invaluable for understanding the basis of therapy resistance and recurrence and for discovering new vulnerabilities in pancreatic cancer,” said Reya. “Beyond providing new scientific insight, this work can help identify pathways for which clinical grade inhibitors may already exist and can thus be rapidly tested in pancreatic cancer.”
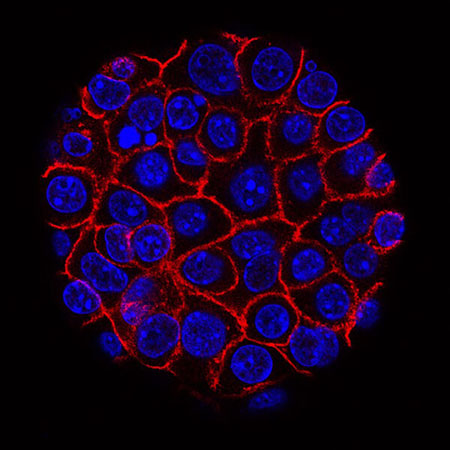
Pancreatic cancer cells (blue) growing as a sphere encased in membranes (red). Photo credit: National Cancer Institute
In particular, she said the discovery of RORγ‘s role in promoting tumor growth provides a new avenue for research and potential therapies. “One exciting aspect is the possibility that nuclear hormone receptors could represent therapeutic targets for pancreatic cancer. In fact, drugs targeting RORγ have already been developed by several pharmaceutical companies, and are in trials for autoimmune diseases. Our findings suggest that these agents could also be a valuable therapeutic strategy for pancreatic cancer.”
Co-authors include: Nikki K. Lytle, L. Paige Ferguson, Nirakar Rajbhandari, Raymond G. Fox, Michael Hamilton, Tami Gilderman, Lourdes Adriana Esparza, Yutaka Shima and Marcie Kritzik, UC San Diego and Sanford Consortium for Regenerative Medicine; Kathryn Gilroy and Neil Robertson, University of Glasgow; Anagha Deshpande, Aniruddha L. Deshpande, and Peter D. Adams, Sanford Burnham Prebys Medical Discovery Institute; Christian M. Schurch, Stanford University; Wei Lin, Pawan Noel, Daniel Von Hoff and Haiyong Han, Translational Genomics Research Institute; Martin Wartenberg, Inti Zlobec, Micha Eichmann, Jose A. Galvan, and Eva Karamitopoulou, University of Bern; Philipp Spahn and Nathan E. Lewis, UC San Diego and Novo Nordisk Foundation Center for Biosustainability; Randall French, UC San Diego Moores Cancer Center; Kathleen M. Fisch, Roman Sasik, Sara Brin Rosenthal, Trey Ideker, and Andrew M. Lowy, UC San Diego.
Funding for this research came, in part, from the National Institutes of Health (grants T32 GM007752, T32 GM007752, F31 CA206416, UL1TR001442, CA169281, R35 GM119850, CA155620, R35 CA197699 and R01 CA186043), the National Foundation for Cancer Research, the Novo Nordisk Foundation, SU2C-CRUK-Lustgarten Foundation Pancreatic Cancer Dream Team Research Grant and SU2C-Lustgarten Foundation Pancreatic Cancer Collective Grant.
Disclosures: Tannishtha Reya is a consultant for Orphagen Pharmaceuticals, and received travel reimbursement from Entertainment Industry Foundation and Lustgarten Foundation. Daniel Von Hoff serves on the scientific advisory board of Five Prime and as a consultant for Celgene. Some data has been filed as a provisional patent application.
Share This:
You May Also Like
Stay in the Know
Keep up with all the latest from UC San Diego. Subscribe to the newsletter today.



