Altman Clinical and Translational Research Institute Opens First Satellite Clinic in Linda Vista
The new clinic’s state-of-the-art research facilities and its location will expand access to clinical trials focused on pediatric diseases.
Published Date
Article Content
UC San Diego’s Altman Clinical and Translational Research Institute (ACTRI) recently opened its new 1,800-square-foot satellite research clinic in the Linda Vista neighborhood of San Diego. The new facility will expand opportunities for researchers to conduct clinical trials on a myriad of conditions that affect children and adolescents by providing pediatric patients access to cutting-edge therapies.
“We are trying to increase research opportunities for all diseases that impact children and adolescents — everything from diabetes to asthma to infectious diseases,” said ACTRI Director David “Davey” Smith, MD, who is also a professor of medicine and assistant vice chancellor of clinical and translational research.
ACTRI has been supporting clinical studies through the Center for Clinical Research at its main facility on the UC San Diego campus in La Jolla since 2016, facilitating everything from groundbreaking research on type 2 diabetes to rare disease studies. It currently serves thousands of adults and hundreds of children every year.
“Despite our success in La Jolla, there was no special place for children to go, and we really viewed that as a significant unmet need,” said Distinguished Professor of Medicine and Senior Associate Vice Chancellor for Health Sciences Gary S. Firestein, MD, ACTRI’s founding director, who spoke at the clinic’s grand opening.

According to Firestein, the vision for the Linda Vista clinic arose out of ACTRI’s commitment to increasing participation in clinical trials by members of the greater San Diego community, including underserved individuals in need of innovative therapies.
The new clinic came about with the support of Vice Chancellor for Health Sciences John Carethers, MD. In early discussions about the clinic, a priority was placed on finding a way for ACTRI to bring world-class care to pediatric patients — instead of requiring them to come to ACTRI’s clinic in La Jolla — while simultaneously expanding opportunities for faculty and research teams to conduct clinical trials near Rady Children’s Hospital.
“We felt that what would work the best was an actual ACTRI satellite across the street from Rady Children’s Hospital that is completely integrated with the rest of ACTRI and provides the same high-quality services that are essential for safe clinical trials,” said Firestein.
Distinguished Professor of Pediatrics and Neuroscience Gabriel G. Haddad, MD, who also serves as chief scientific officer at Rady Children's Hospital was instrumental in securing space for the Linda Vista Clinic within a short walking distance of the hospital.
“We've known for a long time that access matters: if it takes longer for you to get to your doctor's office, then it's less likely that you will go,” said Smith. “The same thing goes for research opportunities. So by removing some of those barriers, especially for people in communities that have farther to go, less reliable transportation, or fewer resources, this provides easier access and more welcoming opportunities.”
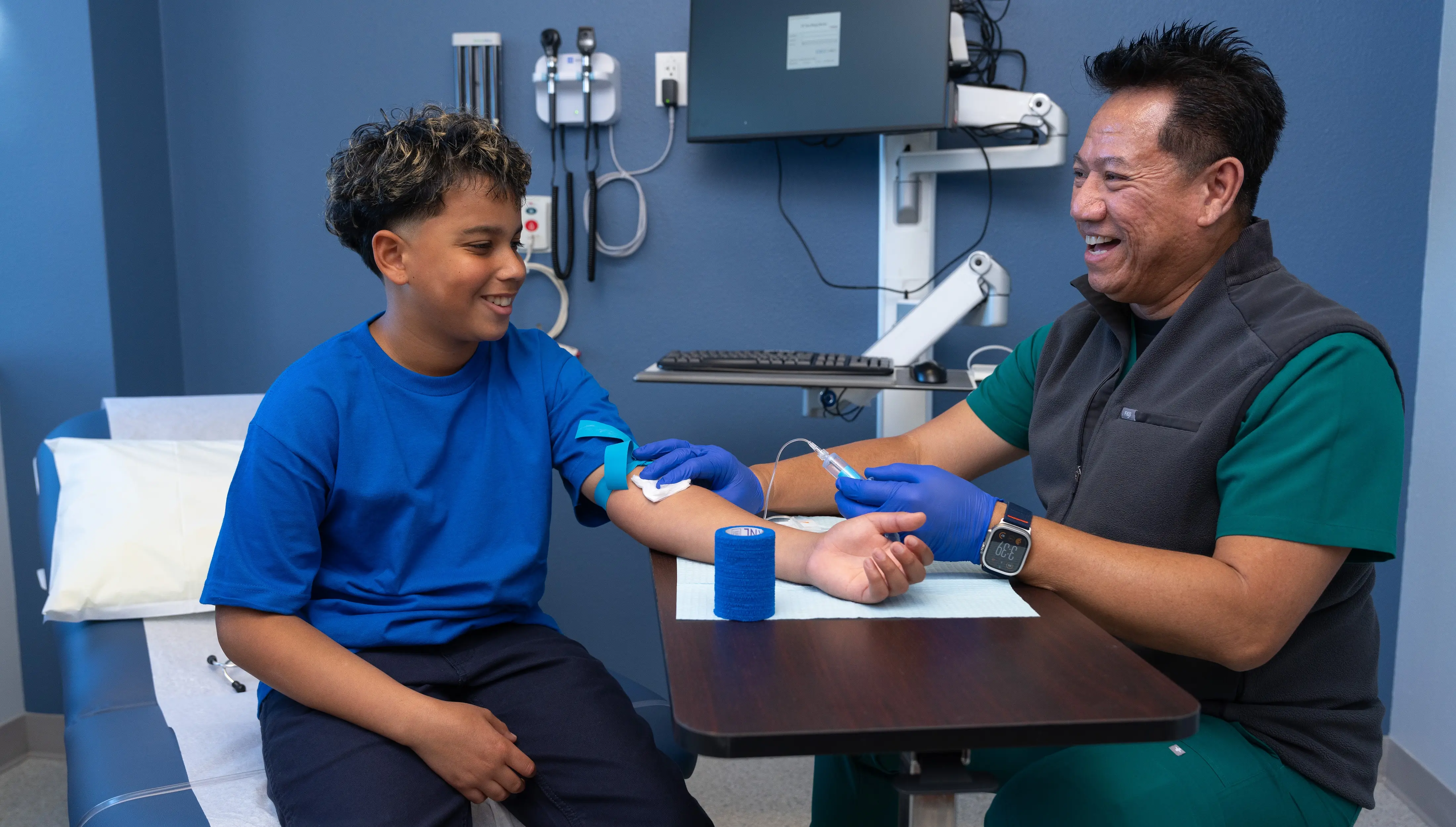

Many diseases affecting children have no approved treatments, according to ACTRI Clinical Director Bernadette Cale, RN. “Clinical research is the last hope for many of these participants. By opening up this satellite facility close to Rady Children’s Hospital, we're going to be able to connect with many more pediatric patients and the faculty at the hospital to provide those lifesaving treatments.”
A one-stop shop
Cale describes the ACTRI Linda Vista clinic as a one-stop shop for researchers, with seven child-friendly exam rooms to meet with study participants — including one for medical procedures and another for cognitive testing — plus a lab and office space for teams to use while conducting research there. The clinic employs research coordinators, nursing staff and lab techs who are shared between various studies, making clinical research more cost effective. Likewise, it supplies regulatory support services to ensure that clinical trials are in compliance with safety standards, project management support, and medical bioinformatics services to analyze data from clinical trials.


Several trials are already underway at the new clinic. One involves pediatric patients with myelin oligodendrocyte glycoprotein antibody-associated disease (MOGAD) — a rare autoimmune disorder of the central nervous system that can cause vision loss, seizures and paralysis among other symptoms, and another focuses on Angelman Syndrome, a rare genetic disorder associated with severe intellectual disability, seizures, and microcephaly.
“All people matter, and we want to make sure that we have research that tackles even the rarest of diseases so that everybody can take advantage of opportunities to improve health,” said Smith.
But studying rare diseases can also contribute to discoveries that have an impact beyond the small number of patients that experience them, he says. “You never know where science will lead you.”


The UC San Diego ACTRI Center for Clinical Research at Linda Vista exemplifies one of UC San Diego Health Sciences’ key strategic goals: fostering research collaborations.
“UC San Diego has had a longstanding partnership with Rady Children’s Hospital,” said Vice Chancellor Carethers. “Together, we have the great responsibility of serving the pediatric population with the highest caliber of evidence-based care. Opening the UC San Diego ACTRI Center for Clinical Research at Linda Vista will strengthen that partnership, allowing us to translate innovative research into cutting-edge therapies that will improve children’s health right here in San Diego County and across the globe.”
Researchers interested in conducting clinical research studies at the Linda Vista clinic should contact Bernadette Cale at bcale@health.ucsd.edu.
Share This:
Stay in the Know
Keep up with all the latest from UC San Diego. Subscribe to the newsletter today.



