Making Heads or Tails Out of Phospholipid Synthesis
Breakthrough research adds to the understanding of creating lipids in artificial cells and of the origins and evolution of cellular membranes
Published Date
Article Content

Luping Liu, from the Devaraj Research Group at UC San Diego, holds a sample of water from a natural alkaline source—Mono Lake, California. Photo courtesy of Luping Liu.
Most scientists agree that life on Earth began about 4 billion years ago, but they don’t agree where—on land or in water. They know that about 2 billion years ago, single-celled organisms evolved into complex plants and animals whose membrane-bound cells had a nucleus and separate compartments, called organelles, with specific functions. This marked an important moment in cellular evolution.
According to an article in Scientific American, some experts say the chemistry of a sea-located, “first life” doesn’t add up. With several hypotheses floating around, researchers are racing to replicate the conditions from which life emerged. UC San Diego award-winning Chemical Biologist Neal Devaraj is one of those scientists whose work focuses on understanding how non-living matter assembles to form life, by approaching the problem through the synthesis of artificial cells.
Together with a team of researchers from UC San Diego, UCLA and the University of South Carolina, Devaraj has demonstrated how membrane-formation takes place in water from natural alkaline sources like soda lakes and hydrothermal oceanic vents. Working in the lab and applying both organic and computational chemistry, the researchers were able to form enzyme-free phospholipid membranes in water from natural alkaline sources—Mono Lake in California and the Lost City Hydrothermal Field in the Atlantic Ocean. Their findings are published in Nature Chemistry.
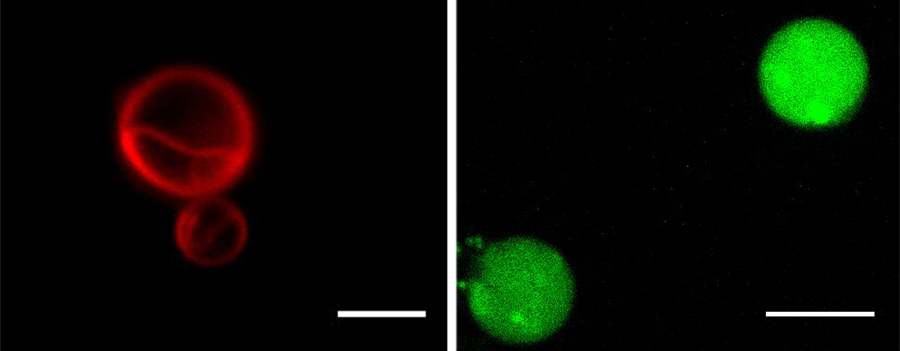
Left: A fluorescence micrograph of vesicles formed in Lost City vent fluid, stained by Nile red dye. Scale bar, 5 μm. Right: Vesicles formed in Mono Lake water (pH = 10) containing the pH indicator dye HPTS. Scale bar, 10 μm. Image created by Luping Liu.
Phospholipids, a class of lipids whose molecule has a “head” containing a phosphate group, and two fatty acid-based “tails,” joined by two ester linkages which hold together glycerol and fatty acids, provide barriers in cellular membranes that protect the cell and make barriers for the organelles within it. They also provide pathways for various substances across cellular membranes.
The chemical description of this work is highly technical—“production of an enzyme-free synthesis of natural diacylphospholipids by transacylation in water, enabled by a combination of ion pairing and self-assembly between lysophospholipids and acyl donors.” Basically this means—on the biology side of their work—the scientists achieved the first efficient, enzyme-free, watery creation of natural phospholipids, offering science new routes for lipid synthesis in artificial cells. To the field of chemistry, their creative strategy combined ion pairing and self-assembly, providing insights on green or sustainable chemistry.
The research results show that phospholipid membranes generated in alkaline water sources are capable of separating charged molecules for hours. Commenting on the work Devaraj said, "It is tempting to speculate that similar phenomena may have occurred in the early origin of membranes, perhaps as alkaline hydrothermal vent water was diluted in the more acidic water of the Hadean ocean.” The Hadean Eon (about 4.6 to 4.0 billion years ago), is when the Earth's seas and atmosphere were developing.
According to Luping Liu, lead author of the published research paper, it was unclear whether natural phospholipid membranes could be formed from simple precursors in water without the advanced enzymatic machinery.
“In general, this work was initiated and motivated by the curiosity for nature, and we solve this challenge by using the principles of molecular interactions inspired by nature,” she said.
Liu also said the team’s findings present a fundamental progress in synthetic lipid study. “Now, we want to extend the application of this creative synthetic strategy of the combination of ion pairing and self-assembly to synthetic biology.”
Support for this work was provided by the National Science Foundation (grant nos. CHE-1254611, CHE-1764328 and OCE-1536702) and the National Institutes of Health (grant nos. DP2DK111801 and R01 GM109078). Computation time was provided by the UCLA Institute for Digital Research and Education (IDRE).
Share This:
Stay in the Know
Keep up with all the latest from UC San Diego. Subscribe to the newsletter today.