Hungry for Love: Gut Molecule Discovered that Flips the Feeding-to-Mating Switch
Nature report uncovers the Dh31 signaling molecule, opening a new paradigm of gut-to-brain communication
Published Date
Article Content
On Valentine’s Day, couples all over the world will enjoy romantic dinners to celebrate love and relationships. The association between nutrition and mating is not unique to humans but is reflected across species throughout the animal kingdom. However, direct physiological links connecting food consumption and reproductive behaviors were not suspected until recently.
Scientists from the University of California San Diego studying fruit flies report that a molecule released from the gut after a meal switches their focus from eating to mating. Publishing Feb. 9 in the journal Nature, the researchers found that protein-rich food triggers the release of diuretic hormone 31, or “Dh31,” a signaling molecule discovered to be released from the fly’s gastrointestinal tract. Their identification of Dh31, a neuropeptide considered to be a type of chemical messenger, opens the door to the largely unexplored area of gut-to-brain communication beyond feeding behavior.
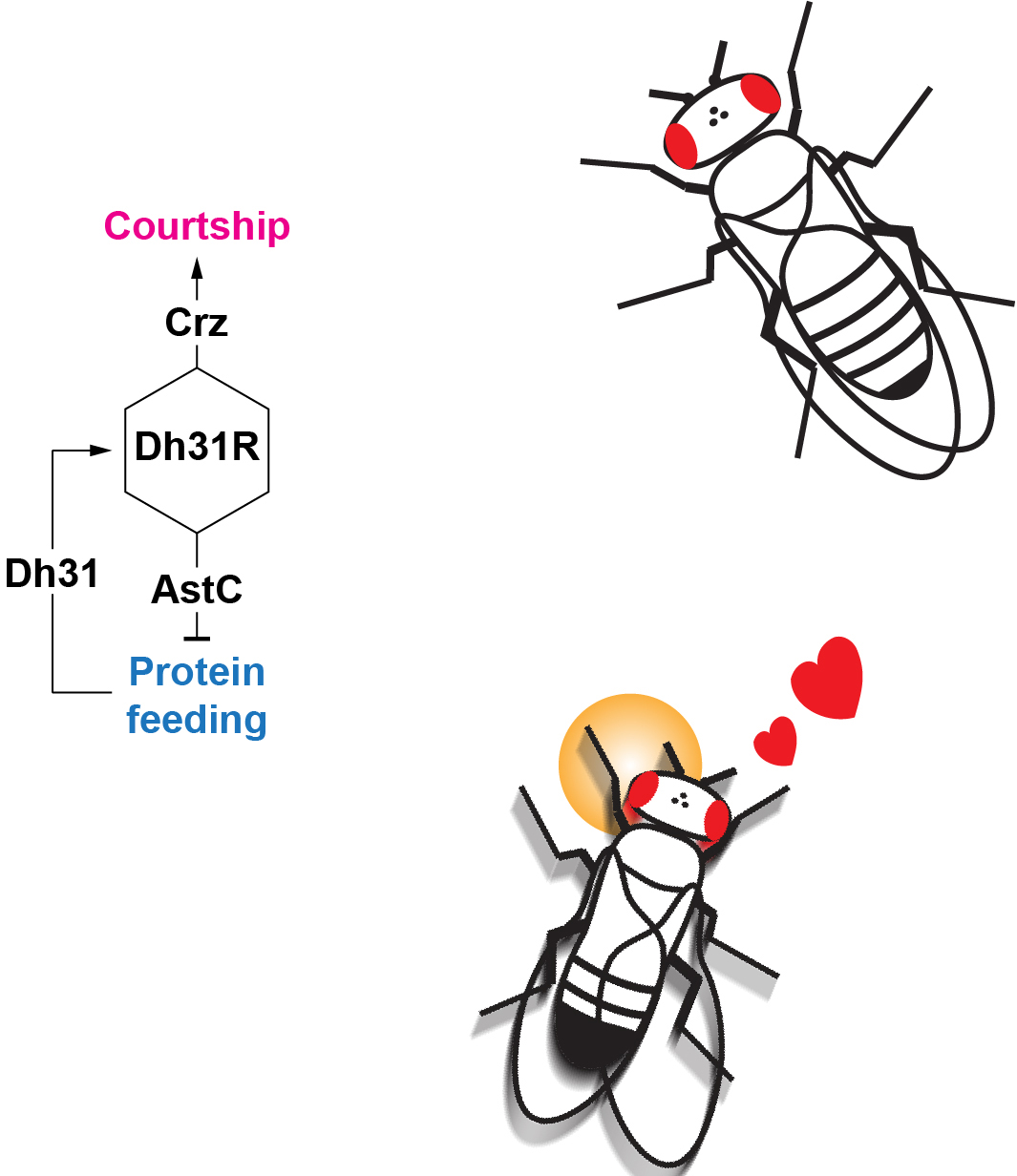
Male fruit flies rapidly switch from feeding to courtship behavior after consuming protein-rich food. Amino acids trigger the release of the neuropeptide Dh31 from the gut, which in turn activates Dh31R-expressing neurons in the brain to terminate feeding and initiate courtship through two parallel circuits.
“We found the transition from feeding to mating and were very surprised that a single molecule would have such a profound influence on behavior decisions,” said Jing Wang, a professor of Neurobiology in the UC San Diego Division of Biological Sciences and the study’s senior author. “Our study provides a mechanistic explanation of how the Dh31 neuropeptide acts on the brain to change the motivational drive of two evolutionarily critical behaviors.”
In investigating the question of how animals change from a feeding-focused behavior to a mating mindset, UC San Diego researchers and their colleagues at UC Santa Cruz used several methods to arrive at their Dh31 molecule discovery, including genetics, three-photon microscopy and fluorescence imaging.
To confirm their finding, the researchers performed genetics experiments in which Dh31 was knocked out of fruit flies. In these cases, the flies kept feeding and held off mating behavior. In other experiments they activated Dh31 and found the flies rapidly flipped to courtship. Instead of the brain, an expected area for the molecule’s release, they found Dh31 originated in the fly’s gut.
“These results indicate that Dh31 is a signaling molecule that reorders the priority of these two contending behaviors: feeding over courtship in the absence of Dh31 and courtship over feeding when Dh31 is released from the gut,” said Wang.
The researchers further described parallels to Dh31’s function as a signaling molecule. Orexin, a neuropeptide molecule, has proven to play a similar role in mammals in the transition from wakefulness to sleep patterns, including rapid eye movement (REM) sleep and non-REM sleep.
These explorations provide insight into decision-making processes when animals switch from one behavior that promotes survival—such as nourishment intake—to a different fundamental behavior such as courtship. Wang said their findings only scratch the surface of understanding how gut hormones function beyond feeding. Future work will investigate how microbiomes factor into gut-to-brain communication.
“This work embodies a multidisciplinary approach to understand behavioral prioritization at multiple levels, from molecules to neurons and circuit function,” said Wang. “This line of work provides us with an empirical paradigm to study the hierarchical organization of different need-based behaviors, a framework established by Abraham Maslow 80 years ago to explain the orderly transition of human behaviors.”
The scientists who contributed to the study include: Hui-Hao Lin, Meihua Christina Kuang, Imran Hossain, Yinan Xuan, Laura Beebe (graduate student), Andrew Shepherd, Marco Rolandi and Jing Wang.
The confocal imaging used in the study was supported by the National Institute of Neurological Disorders and Stroke (P30 NS047101; UC San Diego Neuroscience Microscopy Imaging Core). Funding to support the research was provided by National Science Foundation (1429810) and National Institutes of Health (R01DK127516, R01DK092640, R01DC009597 and K99DC016338).
— With contributions from Susy Kim
Share This:
Stay in the Know
Keep up with all the latest from UC San Diego. Subscribe to the newsletter today.



